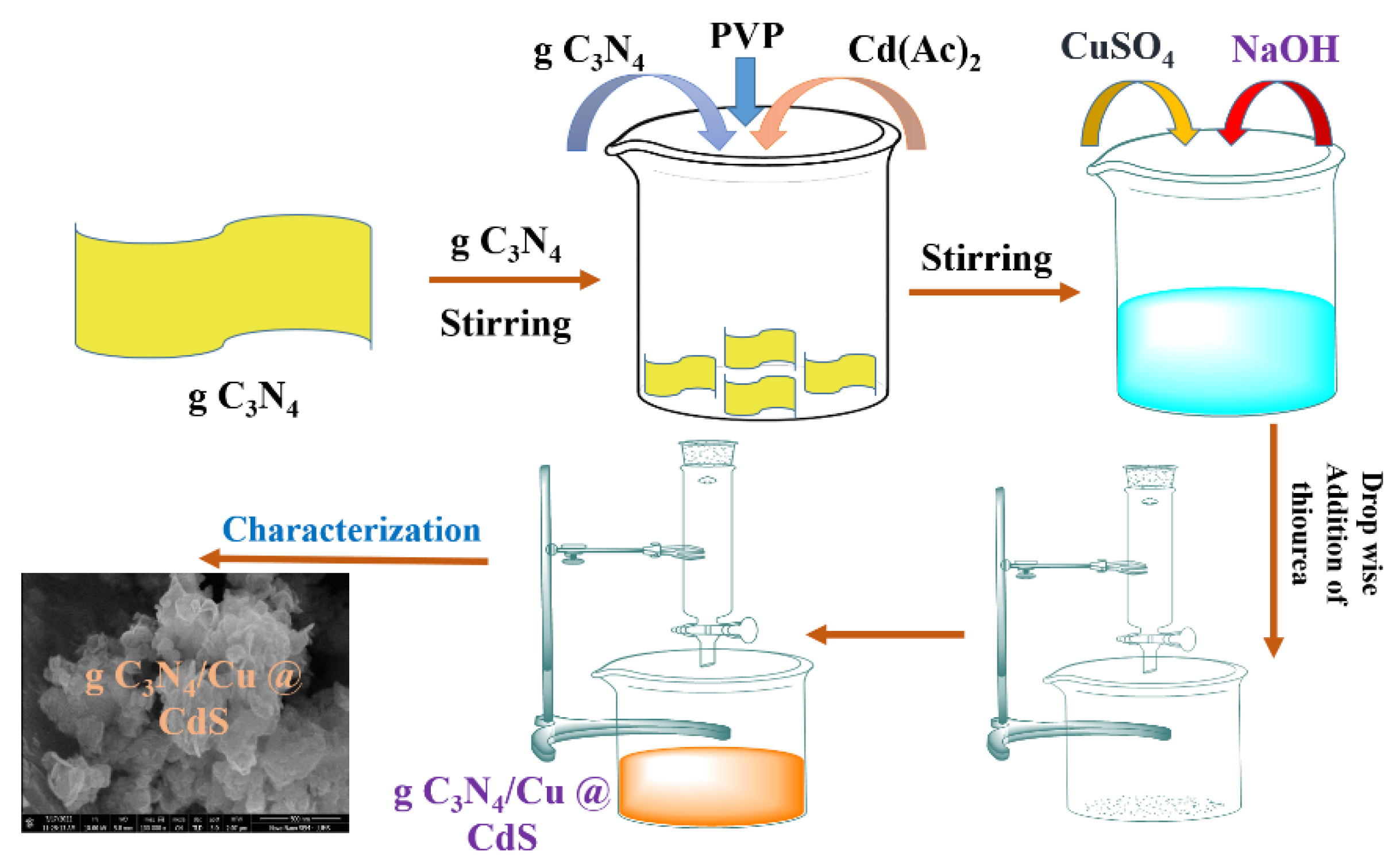
Toxics | Free Full-Text | Fabrication of a Ternary Nanocomposite g-C3N4/Cu@ CdS with Superior Charge Separation for Removal of Organic Pollutants and Bacterial Disinfection from Wastewater under Sunlight Illumination
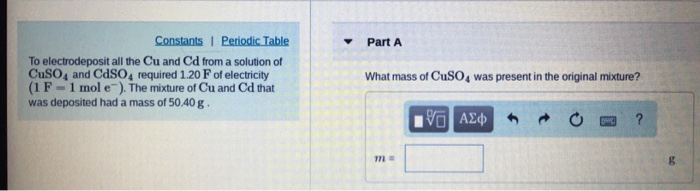
Find the ratio of molar accounts of H2SO4 needed to preciptise metal ions from 20ml each of 1M cd(NO3)2 and 0.5 M cuso4??

Catalytic Activities of CuSO4/Al2O3 in Dehydrogenation of Arenes by Dioxygen | The Journal of Organic Chemistry

Find the ratioof molar amount of H2S needed to precipitise metal ions from 20ml each of 1M Cd(NO3)2 and 0.5M CuSO4 - kqs9srzz

KNR Copper Sulphate Powder - 900 gm Pure Neela thotha, Cuso4 Powder : Amazon.in: Industrial & Scientific

China Copper Sulfate Monohydrate and Pentahydrate Blue Powder Blue Copperas CuSO4 Animal Feed Additive Manufacturer and Exporter | Sustar

China Copper Sulfate Monohydrate and Pentahydrate Blue Powder Blue Copperas CuSO4 Animal Feed Additive Manufacturer and Exporter | Sustar

SOLVED: In which reaction is precipitation occurring? A. MgCl2(aq) + CuSO4(aq) â†' CuCl2(aq) + MgSO4(aq) B. CdSO4(aq) + K2S(aq) â†' CdS(s) + K2SO4(aq) C. NaOH(aq) + NH4Cl(aq) â†' NaCl(aq) + NH4OH(aq) D.











![Copper Sulfate Pentahydrate [CuSO4.5H2O] [CAS_7758-99-8] Fine Crystals – Wintersun Chemical Copper Sulfate Pentahydrate [CuSO4.5H2O] [CAS_7758-99-8] Fine Crystals – Wintersun Chemical](https://www.wintersunchemical.com/cdn/shop/products/03-030-2_1024x1024.jpg?v=1595287760)