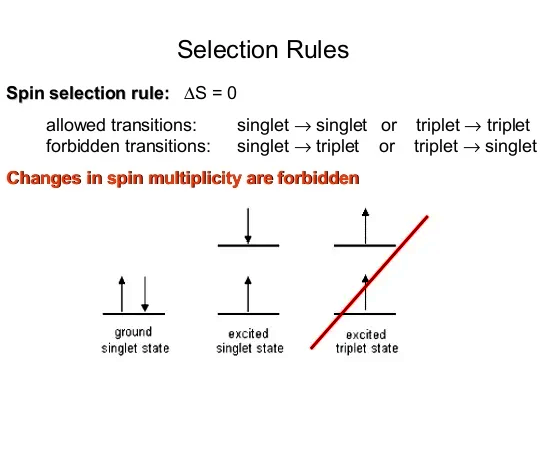
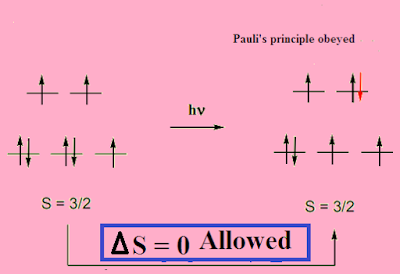
Spin-selective OSE. (A) Optical selection rule for the lowest singlet... | Download Scientific Diagram
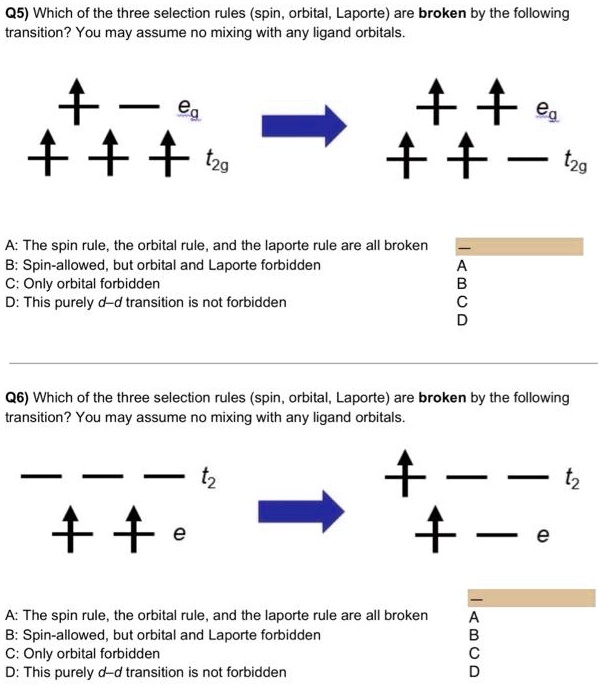
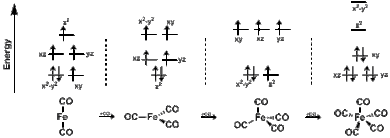
SOLVED: 05) Which of the three selection rules (spin, orbital, Laporte) are broken by the following transition? You may assume no mixing with any ligand orbitals. eg eg t2g t2g A: The

selection rules for d-d transition in hindi, spin selection rule, laporte's selection rule - YouTube

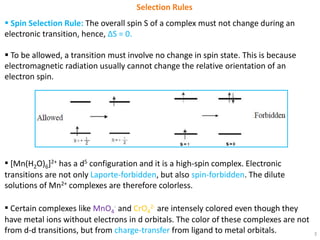
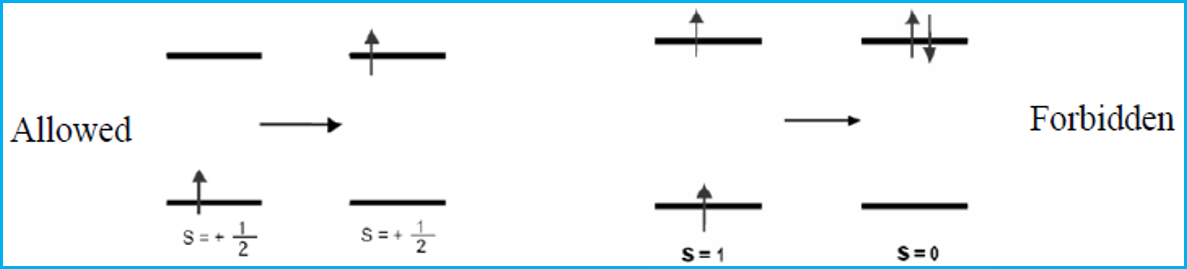
Selection rules.pptx - Selection Rules Some transitions are not allowed does not mean that such a transition will never occur but that it is | Course Hero
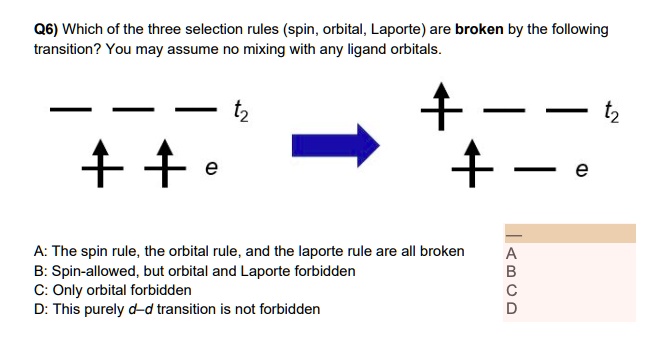
SOLVED: Q6) Which of the three selection rules (spin, orbital, Laporte) are broken by the following transition? You may assume no mixing with any ligand orbitals. A: The spin rule, the orbital

Nonlinear optical selection rule based on valley-exciton locking in monolayer ws2 | Light: Science & Applications
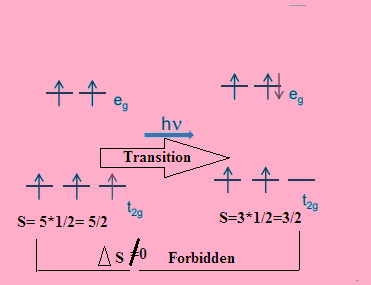
![SOLVED: Explain why an electronic transition for [Mn(H2O)a]2+ is high-spin spin-forbidden, but for H.S. [Co(H2O)6]2+ is spin-allowed. [Mn(H2O)6]2+ is high-spin d5 Mn(II). The transition from t2g to eg orbital is impossible without SOLVED: Explain why an electronic transition for [Mn(H2O)a]2+ is high-spin spin-forbidden, but for H.S. [Co(H2O)6]2+ is spin-allowed. [Mn(H2O)6]2+ is high-spin d5 Mn(II). The transition from t2g to eg orbital is impossible without](https://cdn.numerade.com/ask_images/45883d51a0284fb0993eae22b7c0e0f7.jpg)